Étude lésionnelle et bactériologique des infections du sac vitellin (omphalites) chez les poussins chair, Nord-Est de la Tunisie
Résumé
La présente étude a un triple objectif: d’abord pour estimer la prévalence de l’infection du sac vitellin chez le poulet de chair dans les élevages du Nord-Est de la Tunisie. Le second objectif est d’identifier les principales bactéries responsables de ce problème. Enfin, nous visons l’étude de la sensibilité des souches isolées vis-à-vis des principales molécules d’antibiotiques utilisées sur le terrain. Un total de 170 poussins (3-8 jours d’âge) est autopsié. Les sacs vitellins suspects sont prélevés moyennant des écouvillons stériles envoyés au laboratoire en vue des analyses bactériologiques et des antibiogrammes. La prévalence globale des omphalites est de 55,3% (94 poussins). Une différence statistiquement significative est notée selon les tranches d’âge, avec une plus grande prévalence observée chez les oiseaux âgés de 5 jours (76%). Les analyses bactériologiques ont révélé que 71,3% (67 poussins) des animaux montraient une infection bactérienne unique. E. coli est la bactérie la plus isolées (74,5%), suivie de Klebsiella spp. (31,9%), Salmonella spp. (10,6%), Proteus spp. (9,6%) et Staphylococcus aureus (2,1%). Une corrélation élevée est notée entre la mortalité des poussins et l’infection par E. coli, Klebsiella et Proteus. Les isolats testés (109 isolats) vis-à-vis de 12 antibiotiques montrent une sensibilité importante à la streptomycine, la colistine, l’amoxicilline et l’amoxicilline-acide clavulanique. Une résistance élevée est notée vis-à-vis de l’enrofloxacine. Une multi-résistance est détectée chez 38 souches (34,9%) isolées.
Mots clefs: Infection du sac vitellin, Bactériologie, Multi-résistance aux antibiotiques, poulet de chair, Tunisie
Téléchargements
Introduction
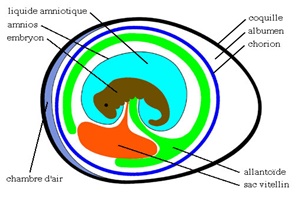
During incubation, the extra-embryonic membranes encircle the yolk reserve and form the yolk sac, which is attached to the intestine via a small canal. Just before hatching, the sac is internalized and moves from the cavity of the egg to the abdominal cavity of the future chick, as an extension of the intestine. This yolk reserve represents 20-25% of the live weight of the chick at birth, then resorbs during the first week and will have a negligible size (Ramnoff, 1960) (in Khan et al. 2004).
However, several factors contribute to the delay of yolk resorption resulting in its retention. Bacterial infections remain among the most frequently factors involved in this health problem. The composition of yolk, very rich in water and lipid promotes the rapid growth of pathogens. In addition, the body temperature of the chick, higher than that of egg incubation, is an ideal factor for bacteria multiplication. Therefore, yolk retention, whatever its cause, leads often to infection of the yolk sac (Khan et al., 2004) which represents the main cause of chick mortality during the first week post hatching (Rai et al., 2005; Yassin et al., 2009). Consequent mortality ranging from 1.7% to 8.6% would cause high economic losses (Awobajo et al., 2007).
Different bacterial species are responsible of yolk sac infection/omphalitis in chicks (Noy et al., 1996; Cox et al. 2005:15; Husseinn et al., 2008; Amare et al. 2013; Jalob et al., 2015; Abdel-Tawab et al., 2016). Infection of the yolk sac can occur through the unhealed umbilicus or the blood stream. In the two cases, infection causes delayed yolk resorption and fatal septicemia of the chick in severe cases.
Proteus spp., Enterobacter spp., Pseudomonas spp., Klebsiella spp., Staphylococcus spp., Streptococcus spp., Clostridium spp., Bacillus spp. and Enterococcus spp. were the main bacteria that have been isolated from yolk sac infections in different region of the world (Rehman et al., 1996; Rad et al., 2003; Cortés et al., 2004; Deeming, 2005; Mondal & Sahoo, 2014; Jalob et al., 2015). Aspergillus fumigatus was also isolated from infected yolk sac (Cortes et al., 2005). Nevertheless, Escherichia coli and Salmonella spp. were frequently the main pathogens isolated from yolk sac infection and omphalitis cases (Shivaprasad 2000; Khalil & El-Shamy, 2012).
In Tunisia, investigations on yolk sac persistence and infections do not receive sufficient attention. Until now, no precise scientific research was published on this topic. Therefore, the aims of this article were to assess the prevalence of yolk sac infections, to identify the causative bacteria and to study the antimicrobial susceptibility of the isolated germs.
Materials and methods
Region of the study and animals
This study was carried out on 170 chicks (120 dead animals and 50 live sick animals) aged from 3 to 8 days. Animals were sampled between September 2014 and July 2017, from 15 broiler flocks located in the north (governorates of Ariana, Bizerte, Tunis, Manouba and Nabeul) of Tunisia. The capacity of farms ranged from 5000 to 18000 birds per flock.
All flocks apply a vaccination program against infectious bronchitis, Newcastle disease and infectious bursal disease.
Necropsy examination
All chicks were autopsied in the avian clinic of the National Veterinary School of Tunisia. Live birds were examined before being euthanized by cervical dislocation followed by bleeding. A meticulous postmortem examination revealed the main lesions in different organs, with special reference to the yolk sac. The lesions allowed us to establish the diagnostic hypotheses.
Sample collection
After necropsy, yolk sac with gross lesions were collected aseptically using sterile plain swabs. The collected samples were labeled, packed and transported under respect of the cold chain principles, to the laboratory.
Bacteriological analysis and antimicrobial susceptibility study
Samples issued from flocks located in the north of Tunisia were analyzed in the microbiology laboratory of the National Veterinary School of Sidi Thabet. Samples collected from flocks located in the region of Sahel were sent to the Regional Center of Veterinary Research of Sousse.
The antimicrobial susceptibility tests were made according to the conventional technique of disk diffusion method, according to the recommendations of the antimicrobial susceptibility committee (French Society of Microbiology). The choice of antibiotics was based on the most frequently used molecules in the field. Results were interpreted directly from bacteriological analysis reports.
Statistical analysis
The results were managed in Microsoft Excel (version 2013). Chi-square test was employed in order to statistically determine if there is any significant difference between studied groups. Correlation test was also employed to assess the association between the chick mortality and the bacterial isolates. In all the analyses p < 0.05 was set for significance.
Results
Prevalence of yolk sac infection
The prevalence of yolk sac infections is indicated in Table 1. Yolk sac infection (YSI) was described in 94 chicks (55.3%), with a higher prevalence observed in 5 days birds (76%), followed by birds at the age of 4 days (66.7%), 6 days (65.4%), 3 days (45.45%), 7 days (41.7%) and 8 days (25%). Difference of YSI prevalence between age groups of animals was statistically significant (p<0.05).
Table 1: Prevalence of yolk sac infection in chicks according to the age (n=170)

Gross pathology
Clinical examination of diseased chicks shows depression, diarrhea and abdominal distension. The presence of a blackish navel button was observed in several birds.
Post-mortem exam reveals an unabsorbed yolk sac, as main lesion observed. Yolk sac appeared congested with a changed content color (greenish yellow or brownish) and consistency of yolk fluid (aqueous, viscous or "caseous") and a thickening of the yolk channel. Putrefactive odor was noted in infected yolk sacs. Subcutaneous blood vessels were dilated and engorged with blood.
In chicks older than 4 days, lesions of systemic infection were observed, as pericarditis, enteritis, enlarged and congested liver and sero-hemorrhagic peritonitis.
Bacteriological analysis
All chicks with infected yolk sac give positive culture, of which, 67 chicks (71.28%) revealed single bacterial species infection whereas, mixed bacterial infections were detected in 27 samples (28.72%).
Out of 94 positive chicks with yolk sac infection, E. coli represents the most important isolated bacteria. This pathogen was detected in 70 birds (74.47%), followed by Klebsiella spp. recovered from 30 chicks (31.91%), Salmonella spp. from 10 birds (10.64%), Proteus spp. in 9 chicks (9.57%) and Staphylococcus aureus in only 2 samples (2.13%).
Results of bacterial analysis were summarized in Table 2. The study of isolation results showed that 121 bacterial strains was interpreted. They were represented mainly by E. coli (70 isolates; 57.85%), followed by Klebsiella spp. (30 isolates; 24.8%), Salmonella spp. (10 isolates; 8.26%), Proteus spp. (9 isolates; 7.44%) and Staphylococcus aureus (2 isolates; 1.65%).
The results of Pearson correlation test revealed a high positive correlation between the chick mortality from 3 to 8 days and E. coli infection (r = 0.811). Secondarily, mortality of chicks correlates with Klebsiella spp. (r = 0.735), followed by Proteus spp. infection (r = 0.622). However, a lower correlation was observed between Salmonella spp. infection (Pearson’s r = 0.434) of yolk sac and bird’s mortality during the few days of age in this study. Finally, a low negative correlation was calculated between chick mortality and Staphylococcus aureus infection (r = -0.222).
Antimicrobial sensitivity test
Antimicrobial sensitivity test targeted isolates of E. coli, Klebsiella spp. and Salmonella spp. Twelve antibiotic agents were tested. The detailed results are summarized in Table 3. In general, the overall susceptibility of bacterial isolates varied from 20.95% (enrofloxacin) to 87.64% (streptomycin), depending on the used antibiotic (Figure 1).
E. coli was found to be highly sensitive to ampicillin, amoxicillin-clavulanic acid and strepromycin, as following: 91.94%, 85.71% and 87.93%, respectively. However, very high resistance was detected to enrofloxacin (88.57%), followed by tetracycline (71.43%), gentamicin (66.67%) and florfenicol (60%).
Table 2: Correlation between mortality due to yolk sac infections and different bacterial isolates according to the age

Table 3: Sensitivity of E. coli, Klebsiella spp. and Salmonella spp. isolates to different antibiotic agents

Figure 1: Antimicrobial sensitivity of E. coli. Klebsiella spp. and Salmonella spp. isolates from chicks with yolk sac infections

The analysis of Klebsiella spp. sensitivity indicates high susceptibility to colistin (93.33%), followed by streptomycin (90.91%), gentamicin (89.29%) and trimethoprim-sulfamethoxazol (87.50%). High resistance was detected to enrofloxacin (53.85%) and to amoxicillin-clavulanic acid (50%). Salmonella spp. was found to be highly sensitive to colistine (88.89%). Secondarily, the results demonstrated a susceptibility of isolates to streptomycin (77.78%), gentamicin (66.67%) and doxycycline (66.67%). On the other hand, resistance was notable to enrofloxacin (77.78%), followed by amoxicillin-clavulanic acid, amoxicillin, tetracycline and florfenicol, with the same rate (66.66%).
Multidrug resistant (MDR) isolates were detected in this study. Out of 109 tested isolates, 38 bacteria isolates (34.86%) showed a resistance to more than three antibiotics of three different antimicrobial families. These MDR germs were represented by 26 E. coli (68.42%) isolates, 10 Klebsiella spp. (26.31%) isolates and 2 Salmonella spp. (5.26%) isolates.
Discussion
Yolk sac infection is an economically important and frequent affection, usually observed during the first week of life, inducing high mortality and low performances. Studying navel condition in chicks at hatch, Fasenko & O’Dea (2008) indicate a reduction in production efficiency of birds with navel buttons. These chicks had also higher mortality over the entire production period compared to broilers with leaky or normal navels. The incidence of YSI is variable from 5% to 30% depending on published studies. A higher prevalence is noted during cold season (winter, autumn) (Khan et al., 2004).
Overall prevalence of YSI in this study was 55.29%, which is higher than result of Amare et al. (2013) who published a prevalence of 33.1% (96 positive birds / 290 examined). A higher incidence was observed in 5 days old chicks (76%), followed by birds aged of 4 days (66.67%) and 6 days (65.38%) (p<0.05). Our findings are in agreement with results of Amare et al. (2013). Many factors promote early YSI such as poor hatchery hygienic conditions, poor transport conditions and delay in the transport from the hatchery to the farm. The delay of feeding post hatching is one of important causes of yolk sac retention, which consists a good culture medium for bacteria. This suggests that yolk transfer into the intestine may be accelerated by early feeding, causing intestinal motility (Noy & Skalan, 1997; Murakami et al., 1998). Retained yolk sac will exacerbate the deprivation of nutrient and maternal derived antibodies, the development and spread of bacteria and the adsorption of toxins. On the other hand, unabsorbed yolk sac could be a source of Salmonella Enteritidis and Campylobacter spp., which will be excreted in droppings under stress condition, and spread to the carcass of future broilers (Desmidt et al., 1997; Cox et al., 2005).
Khan et al. (2002) studied the pathogenesis of yolk retention in broiler chicks. They inoculated E. coli into the yolk of the chicks and observed that yolk sac infection led to decrease in rate of yolk absorption. They also observed that the body weight was significantly lower and the weight of the unabsorbed yolk was higher in infected chicks than in control chicks. Rai et al. (2003) also observed the same effects in experimental yolk sac infection with Staphylococcus aureus.
According to the passive protection, yolk sac infections affect maternal immunity in chicks. In this way, bacterial contamination can alter protein structures in the yolk. Experimental inoculation of Enterococcus faecalis or E. coli into the yolk causes significant decrease in Newcastle disease virus antibody absorption (Sander et al., 1998; Shah et al., 2004).
The diseased chicks examined in this study, appeared depressed with distended abdomens and signs of diarrhea and dehydration. Signs of omphalitis were observed, as their navel was markedly thickened with usually a brown button, result of unhealed navel. Retained yolk sac was externally palpable. At necropsy exam, described lesions were similar to what was reported by other authors (Shaw & Halvorson, 1993; Khan et al., 2004; Amare et al., 2013; Jalob et al., 2015).
In this study, Gram-negative bacteria isolates predominated in yolk sac infections. Out of 94 culture positive of YSI chicks, single infection was most frequent (67 chicks; 71.28%). As previously reported (Rosario et al., 2005; Iqbal et al., 2006; Husseinn et al., 2008:48-56; Amare et al., 2013), E. coli was the predominant isolate in this study (70 chiks; 74.47%), followed by Klebsiella spp. (30 chicks; 31.91%), Salmonella spp. (10 chiks; 10.64%), Proteus spp. (9 chicks; 9.57%) and Staphylococcus aureus (2 chiks; 2.13%). A high positive correlation (r = 0.811) was observed between the chick mortality from 3 to 8 days and E. coli infection.
Our findings agree with reports of Mosqueda and Lucio (1985) (in Husseinn et al. 2008:48-56), Cortes et al. (2004) and Amare et al. (2013). However, Jalob et al. (2015), isolated more Streptococcus spp. than E. coli from yolk sac infection cases in Egypt. Similarly, Rad et al. (2003) identified mainly Gram-positive bacteria (Streptococcus species, Staphylococcus species, Bacillus cereus, Clostridium perfringens) in broilers flocks in Iran.
This study revealed the presence of Klebsiella spp. in 30 affected chicks (31.91%). This prevalence was higher than prevalence mentioned by Iqbal et al. (2006) (1.79%), Jalob et al. (2015) (3.1%) and Khalil & El-Shamy (2012) (4.6%). However, there was no isolation of Klebsiella spp. from yolk sac infection cases in other reports (Nasrin et al., 2012; Amare et al., 2013; Abdel-Tawab et al., 2016).
Mixed bacterial infections, detected in 27 cases (28.72%), were due to E. coli together with Proteus spp or with Klebsiella spp. Our findings do not agree with the results of Amare et al. (2013), who mentioned predominant mixed infections of yolk sac compared to single infection.
Globally, antimicrobial sensitivity test interesting twelve antibiotic agents revealed overall highly susceptibility to streptomycin (87.64%), followed by colistin (79.82%), amoxicillin (77.06%) and amoxicillin-clavulanic-acid (75.79%). Our results are partially in agreement with the findings of Amare et al. (2013).
Most of E. coli tested isolates were resistant to enrofloxacin, tetracycline, gentamicin and florfenicol. Our findings are partially similar to results of Das et al. (2017) concerning tetracycline. However, they are not in agreement with reports of Abdel-Tawab et al. (2016), who mentioned high resistance of E. coli isolates to amoxicillin.
The analysis of Klebsiella spp. sensitivity indicates high sensitivity to colistin (93.33%), followed by streptomycin (90.91%), gentamicin (89.29%) and trimethoprim-sulfamethoxazol (87.50%). These findings are partially in agreement with those of Iqbal et al. (2006), who mentioned resistance of the whole Klebsiella spp. isolates to trimethoprim-sulfamethoxazol and to streptomycin.
The analysis of Salmonella spp. sensitivity indicates high susceptibility to colistin and high resistance to enrofloxacin. These findings do not agree with previous reports of Nasrin et al. (2012), who founded higher resistance of Salmonella isolates to tetracycline, erythromycin and nalidixic-acid. In addition, Abdel-Tawab et al. (2016) mentioned a higher resistance of Salmonella to amoxicillin.
Multidrug resistant (MDR) isolates were detected in this study, as reported in many previous reports (Iqbal et al., 2006; Das et al., 2017). This emerging problem is alarming as this antimicrobial resistance may represent a sanitary risk to animal and human health. Several factors explain antimicrobial resistance in poultry productions: abusive use of antibiotics, failed biosecurity measures, lack of official controls, existence of illegal market of chicks and drugs and use of antibiotics without veterinary prescription.
Further sensitization and guidance of poultry professional are required for the prevention of early infections and optimal profitability.
Conclusion
The results of the present study indicate that YSI is a great threat to the broiler’s flocks in the northeast of Tunisia. However, this problem is underestimated, compared to other infectious and parasitic diseases in the region. Despite the susceptibility of isolated bacteria to many antibiotics which may be effective in the treatment of omphalitis, the emergence of multidrug-resistant germs is a real danger to animal and human health. Further investigations are needed to better control risk factors and reduce the frequent use of antibiotics during the first week of age of chickens. The real economic impact of yolk sac infections on a wider scale also needs to be studied in order to introduce more rational preventive measures.
References
Abdel-Tawab A.A., Nasef S.A. and Ibrahim O.A. (2016). Bacteriological and molecular studies on bacteria causing omphalitis in chicks with regard to disinfectant resistance. Global Veterinaria 17(6): 539-545. doi: 10.5829/idosi.gv.2016.539.545
Amare A., Mohammed Amin A., Shiferaw A., Nazir S. and Negussie H. (2013). Yolk sac infection (omphalitis) in Kombolcha poultry farm, Ethiopia. American-Eurasian J. Sci. Res. 8: 10-14.
Awobajo O.K., AKintan Y.M., Igbosanu A.O., Mako A.A. and Olatokunbo O.T. (2007). The mortality rate in the two breeds of broiler on breeding stage. World Applied Science Journal 2(4): 304-308.
Cortés C.R., Téllez Isaías G., López Cuello C., Villaseca-Flores J.M., Anderson R.C. and Eslava Campos C. (2004). Bacterial isolation rate from fertile eggs, hatching eggs, and neonatal broilers with yolk sac infection. Rev. Latinoam Microbiol. 46(1-2): 12-16.
Cortes P.L., Shivaprasad H.L., Kiupel M. and Sentíes-Cué G. (2005). Omphalitis associated with Aspergillus fumigatus in poults. Avian Diseases 49(2): 304-308.
Cox N.A., Richardson L.J., Buhr R.J., Northcutt J.K., Fairchild B.D., Mauldin J.M. and Bailey J.S. (2005). Presence of various naturally occurring bacteria in unabsorbed yolks of six-week-old commercial broilers. Poult. Sci. 84 (Suppl.1): 15.
Das A., Sen A., Dhar P.K., Nath S.K., Ghosh P. and Saifuddin A.K.M. (2017). Isolation of Escherichia coli from the liver and yolk sac of day old chicks with their antibiogram. British Journal of Biomedical and Multidisciplinary Research 1(1): 19-25.
Deeming D.C. (2005). Yolk sac, body dimensions and hatchling quality of ducklings, chicks and poults. British Poultry Science 46(5), 560-564. doi: 10.1080/00071660500255042
Desmidt M., Ducatelle R. and Haesebrouck F. (1997). Pathogenesis of Salmonella Enteritidis phage type four after experimental infection of young chickens. Vet. Microbiol. 56: 99-109.
Fasenko G.M. and O’Dea E.E. (2008). Evaluating broiler growth and mortality in chicks with minor navel conditions at hatching. Poultry Science 87: 594-597. doi:10.3382/ps.2007-00352
Husseinn S.A., Hassan A.H. and Sulaiman R.R. (2008). Bacteriological and pathological study of yolk sac infection in broiler chicks in Sulaimani district. J. Dohuk Univ. 11(1): 48-56.
Iqbal M., Shah I.A., Ali A., Khan M.A. and Jan S. (2006). Prevalence and in vitro antibiogram of bacteria associated with omphalitis in chicks. Pakistan Veterinary Journal 26(2): 94-96.
Jalob Z.K., Farhan W.H., Ibrahiem Z.Y. and Jumaa B.N. (2015). Bacteriological and pathological study of omphalitis in broiler chicks. Kufa Journal For Veterinary Medical Sciences 6(2): 17-26.
Khalil S.A. and El-Shamy E. (2012). Aerobic bacteria associated with omphalitis of chicks. Alex. J. Vet. Science 37(1): 69-77.
Khan K.A., Khan S.A., Aslam A., Rabbani M. and Tipu M.Y. (2004). Factors contributing to yolk retention in poultry: A review. Pakistan Veterinary Journal 24: 46-50.
Khan K.A., Khan S.A., Hamid S., Aslam A. and Rabbani M. (2002). A study on the pathogenesis of yolk retention in broiler chicks. Pakistan Veterinary Journal 22(4): 175-180.
Mondal D. and Sahoo S.K. (2014). Omphalitis in ducklings with Staphylococcus aureus infection. J. Anim. Res. 4(2): 217-222. doi: 10.5958/2277-940X.2014.00008.4
Murakami H., Akiba Y. and Horiguchi M. (1988). Energy and protein utilization in newly hatched broiler chicks: studies on the early nutrition of poultry. Japanese Journal of Zootechnological Science 59: 890-895
Nasrin S., Islam M.A., Khatun M., Akhter L. and Sultana S. (2012). Characterization of bacteria associated with omphalitis in chicks. The Bangladesh Veterinarian 29(2): 63-68.
Noy Y., Uni Z. and Sklan D. (1996). Routes of yolk utilization in the newly hatched chick. British Poultry Science 37: 987-995.
Noy Y. and Skalan D. (1997). Post-hatch development in poultry. Journal of Applied Poultry Research 6: 344-354.
Rad M., Esmailnejad Z. and Keleidari Gh. (2003). Identification of gram-positive bacteria involved in yolk sac infection. (abstract) Acta Vet. Scand. Suppl. 98-2003.
Rai M.F., Khan S.A., Aslam A., Saeed K., Khan K.A. and Shah M.S. (2003). Effect of experimental yolk sac infection with Staphylococcus aureus on immune status of broilers. Pakistan Veterinary Journal 23: 84-90.
Rai M.F., Khan S.A., Aslam A. and Saeed K. (2005). Effect of yolk sac infection in chicken. Avian Poult. Boil. Rev. 16: 87-93.
Rehman R., Rabbani M., Khan S.A. and Saleem C.M. (1996). Pathological aspects of early chick mortality due to bacterial infections. Pakistan Journal Science Research 48(3-4): 101-107.
Rosario C.C., Puente J.L., Verdugo-Rodriguez A., Anderson R.C. and Eslava C.C. (2005). Phenotypic characterization of ipaHþ Escherichia coli strains associated with yolk sac infection. Avian Disease 49: 409-417.
Sander J.E., Willinghan E.M., Wilson J.L. and Thayer S.G. (1998). The effect of inoculating Enterococcus faecalis into the yolk sac on chick quality and maternal antibody absorption. Avian Diseases 42(2): 359-363.
Shah M.S.D., Khan S.A., Aslam A., Rabbani M., Khan K.A. and Rai M.F. (2004). Effect of experimental yolk sac infection with Escherichia coli on immune status of broiler chicks. Pakistan Veterinary Journal 24: 125-128.
Shaw D.P. and Halvorson D.A. (1993). Early chick mortality associated with rupture of the yolk sac. Avian Diseases 37(3): 720-723
Shivaprasad HL. (2000). Fowl typhoid and pullorum disease. Revue Scientifique et Technique 19(2): 405-424.
Yassin H., Velthuis A.G.J., Borjan M. and Van Rilet J. (2009). Field study on broilers first-week mortality. Poultry Science 88: 798-804.
Téléchargements
Publié-e
Numéro
Rubrique
Licence

Revue Marocaine des Sciences Agronomiques et Vétérinaires est mis à disposition selon les termes de la licence Creative Commons Attribution - Pas d’Utilisation Commerciale - Partage dans les Mêmes Conditions 4.0 International.
Fondé(e) sur une œuvre à www.agrimaroc.org.
Les autorisations au-delà du champ de cette licence peuvent être obtenues à www.agrimaroc.org.

